What is the difference between 201 stainless steel and 304 stainless steel?
 What is the difference between 201 st...
What is the difference between 201 st...Copper, bronze, cupronickel and red copper are one of the commonly used non-standard materials, especially brass and bronze.

Brass, bronze (blue-gray or gray-yellow), white copper, and red copper (also known as "red copper" or "red copper") are distinguished by color, among which brass and white copper are very easy to distinguish; red copper is pure copper ( Impurities < 1%), bronze (other alloy components about 5%) are slightly difficult to distinguish. When unoxidized, the color of red copper is brighter than that of bronze, and the bronze is slightly cyan or yellowish dark; after oxidation, the red copper becomes black, and the bronze is turquoise (harmful oxidation of water) or chocolate.

Brass is an alloy composed of copper and zinc. Brass composed of copper and zinc is called ordinary brass. If it is composed of two or more elements, the elements that are often added are lead, tin, aluminum, etc. Various alloys are called special brass.
Red copper is a relatively pure copper, which can generally be regarded as pure copper, but it is not necessarily pure copper, and sometimes a small amount of deoxidizing elements or other elements are added to improve the material and performance, so it is also classified as copper alloy.
Bronze is an alloy of pure copper (red copper) added with tin or lead.
Cupronickel is a copper-based alloy with nickel as the main additive element. The copper-nickel binary alloy is called ordinary cupronickel; the cupronickel alloy with manganese, iron, zinc, aluminum and other elements is called complex cupronickel.

Brass has strong wear resistance, and brass is often used to manufacture valves, water pipes, connecting pipes for internal and external air conditioners, and radiators.
The electrical conductivity and thermal conductivity of red copper are second only to silver, and are widely used in the production of electrical and thermal conductivity equipment. Copper has good corrosion resistance in the atmosphere, seawater and some non-oxidizing acids (hydrochloric acid, dilute sulfuric acid), alkalis, salt solutions and various organic acids (acetic acid, citric acid).
Bronze has the characteristics of low melting point, high hardness, strong plasticity, wear resistance, corrosion resistance, bright color, etc. It is suitable for casting various appliances, mechanical parts, bearings, gears, etc.
Compared with other copper alloys, cupronickel has exceptionally good mechanical and physical properties, good ductility, high hardness, beautiful color, corrosion resistance, and deep-drawing properties. It is widely used in shipbuilding, petrochemical, electrical appliances, instrumentation, medical equipment, daily necessities , handicrafts and other fields, and is also an important resistance and thermocouple alloys.

Brass is a kind of copper containing other alloy components. Its price is cheaper than that of red copper, and its conductivity and plasticity are slightly worse than that of red copper, but its strength and hardness are higher.

Brass is an alloy of copper and zinc. The simplest brass is a copper-zinc binary alloy, called simple brass or ordinary brass, and brass with different mechanical properties can be obtained by changing the zinc content in brass. The higher the zinc content in brass, the higher the strength and the lower the plasticity. The zinc content of brass used in the industry does not exceed 45%. No matter how high the zinc content is, it will cause brittleness and deteriorate the alloy properties. Adding 1% tin to brass can significantly improve the resistance of brass to seawater and marine atmospheric corrosion, so it is called "navy brass". Tin can improve the machinability of brass. Lead brass is what we usually call easy-cut national standard copper. The main purpose of adding lead is to improve machinability and wear resistance, and lead has little effect on the strength of brass. Engraved copper is also a type of lead brass. Most brasses have good color, workability, ductility, and are easy to electroplate or paint.
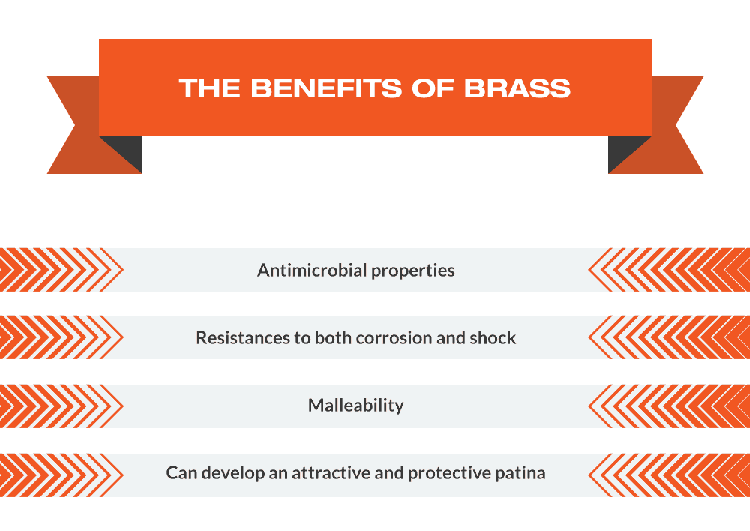
In industry and civil use, different materials are selected according to different use characteristics. Such as wire, requires softer, copper is better. If it is used as a connecting piece, the place where the screw is attached is mostly made of brass.

Originally refers to copper-tin alloys, and later copper alloys other than brass and cupronickel are called bronzes, and are often prefixed with the name of the first main added element before the name of bronze. Tin bronze has good casting properties, anti-friction properties and good mechanical properties, and is suitable for manufacturing bearings, worm gears, gears, etc. Lead bronze is a widely used bearing material for modern engines and grinders. Aluminum bronze has high strength, good wear resistance and corrosion resistance, and is used for casting high-load gears, bushings, marine propellers, etc. Beryllium bronze and phosphor bronze have high elastic limit and good electrical conductivity, and are suitable for the manufacture of precision springs and electrical contact elements. Beryllium bronze is also used to manufacture non-sparking tools used in coal mines and oil depots.

A copper alloy with nickel as the main additive element. The copper-nickel binary alloy is called ordinary cupronickel; the cupronickel alloy with manganese, iron, zinc, aluminum and other elements is called complex cupronickel. Industrial cupronickel is divided into two categories: structural cupronickel and electrical cupronickel. The characteristics of structural cupronickel are good mechanical properties and corrosion resistance, and beautiful color. This cupronickel is widely used in the manufacture of precision machinery, chemical machinery and ship components. Electrical cupronickel generally has good thermoelectric properties. Manganin, constantan and test copper are manganese cupronickel with different manganese content, and are used in the manufacture of precision electrical instruments, varistors, precision resistors, strain gauges, thermocouples, etc.

Pure copper is a purple-red metal, commonly known as "red copper", "red copper" or "red copper". Pure copper is ductile. Pure copper the size of a drop of water can be drawn into filaments up to two kilometers long, or rolled into almost transparent foils larger than a bed. The most valuable property of pure copper is that its electrical conductivity is very good, second only to silver among all metals, so it has become the "protagonist" of the electrical industry. Pure copper is much more widely used than pure iron, and 50% of copper is electrolytically purified to pure copper every year for the electrical industry. The pure copper mentioned here really needs to be very dull, with a copper content of more than 99.95%. Very small amounts of impurities, especially phosphorus, arsenic, aluminum, etc., can greatly reduce the conductivity of copper. Oxygen in copper (a small amount of oxygen is easily mixed in copper smelting) has a great influence on electrical conductivity, and copper used in the electrical industry must generally be oxygen-free copper. In addition, impurities such as lead, antimony, and bismuth will make the crystals of copper unable to combine together, causing hot brittleness and affecting the processing of pure copper. This high-purity pure copper is generally refined by electrolysis: using impure copper (that is, blister copper) as the anode, pure copper as the cathode, and copper sulfate solution as the electrolyte. When the current passes through, the impure copper on the anode gradually melts, and the pure copper gradually precipitates on the cathode. The copper obtained in this way; the purity can reach 99.99%.
 What is the difference between 201 st...
What is the difference between 201 st... Why is 316 stainless steel better tha...
Why is 316 stainless steel better tha... 400 series stainless steel science
40...
400 series stainless steel science
40... How to distinguish the processing tec...
How to distinguish the processing tec... Non-standard design materials of bras...
Non-standard design materials of bras... What type of titanium alloy does Tc4 ...
What type of titanium alloy does Tc4 ...